Researchers Create Self-Replicating Living Robots
Nov 30, 2021 by News Staff / Source
Professor Joshua Bongard, a computer scientist and robotics expert at the University of Vermont, and his colleagues from Tufts University and Harvard University’s Wyss Institute for Biologically Inspired Engineering — the same team that built the first living robots (Xenobots, assembled from cells of a frog species called Xenopus laevis) in 2020 — have discovered that these computer-designed and hand-assembled organisms can swim out into their tiny dish, find single cells, gather hundreds of them together, and assemble ‘baby’ Xenobots inside their Pac-Man-shaped ‘mouth’ — that, a few days later, become new Xenobots that look and move just like themselves; and then these new Xenobots can go out, find cells, and build copies of themselves.
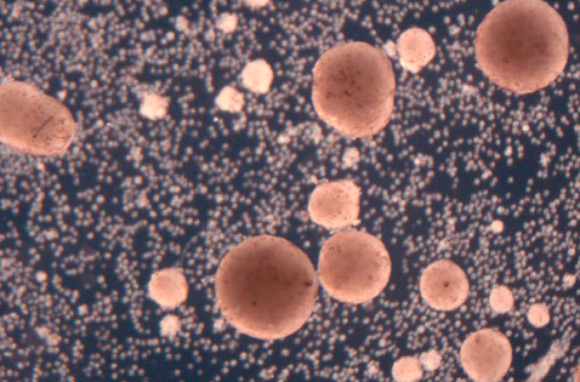
Kriegman et al. show that clusters of cells, if freed from a developing organism, can similarly find and combine loose cells into clusters that look and move like they do, and that this ability does not have to be specifically evolved or introduced by genetic manipulation. Image credit: Kriegman et al., doi: 10.1073/pnas.2112672118.
“In a Xenopus laevis frog, these embryonic cells would develop into skin. They would be sitting on the outside of a tadpole, keeping out pathogens and redistributing mucus,” said Professor Michael Levin, director of the Allen Discovery Center at Tufts University.
“But we’re putting them into a novel context. We’re giving them a chance to reimagine their multicellularity.”
“People have thought for quite a long time that we’ve worked out all the ways that life can reproduce or replicate,” added Dr. Douglas Blackiston, the senior scientist at Tufts University.
“But this is something that’s never been observed before.”
In their earlier experiments, the authors were amazed that Xenobots could be designed to achieve simple tasks.
Now they are stunned that these biological objects will spontaneously replicate.
“We have the full, unaltered frog genome, but it gave no hint that these cells can work together on this new task, of gathering and then compressing separated cells into working self-copies,” Professor Levin said.
“These are frog cells replicating in a way that is very different from how frogs do it. No animal or plant known to science replicates in this way,” added Dr. Sam Kriegman, a postdoctoral researcher at Tuft’s Allen Center and Harvard University’s Wyss Institute for Biologically Inspired Engineering.
https://youtu.be/aBYtBXaxsOw
On its own, the Xenobot parent, made of some 3,000 cells, forms a sphere.
“These can make children but then the system normally dies out after that. It’s very hard, actually, to get the system to keep reproducing. But with an artificial intelligence program working on the Deep Green supercomputer cluster at Vermont Advanced Computing Core, an evolutionary algorithm was able to test billions of body shapes in simulation — triangles, squares, pyramids, starfish — to find ones that allowed the cells to be more effective at the motion-based ‘kinematic replication’ reported in the new research,” Dr. Kriegman said.
“We asked a supercomputer to figure out how to adjust the shape of the initial parents, and the AI came up with some strange designs after months of chugging away, including one that resembled Pac-Man.”
Kinematic replication is well-known at the level of molecules — but it has never been observed before at the scale of whole cells or organisms.
“We’ve discovered that there is this previously unknown space within organisms, or living systems, and it’s a vast space,” Professor Bongard said.
“How do we then go about exploring that space? We found Xenobots that walk. We found Xenobots that swim. And now, in this study, we’ve found Xenobots that kinematically replicate. What else is out there?”
The researchers see promise in the research for advancements toward regenerative medicine.
“If we knew how to tell collections of cells to do what we wanted them to do, ultimately, that’s regenerative medicine — that’s the solution to traumatic injury, birth defects, cancer, and aging,” Professor Levin said.
“All of these different problems are here because we don’t know how to predict and control what groups of cells are going to build. Xenobots are a new platform for teaching us.”
The team’s work was published in the Proceedings of the National Academy of Sciences.
http://www.sci-news.com/biology/xenobots-10317.html?utm_source=feedburner&utm_medium=email
Thanks to: http://www.sci-news.com
Nov 30, 2021 by News Staff / Source
Professor Joshua Bongard, a computer scientist and robotics expert at the University of Vermont, and his colleagues from Tufts University and Harvard University’s Wyss Institute for Biologically Inspired Engineering — the same team that built the first living robots (Xenobots, assembled from cells of a frog species called Xenopus laevis) in 2020 — have discovered that these computer-designed and hand-assembled organisms can swim out into their tiny dish, find single cells, gather hundreds of them together, and assemble ‘baby’ Xenobots inside their Pac-Man-shaped ‘mouth’ — that, a few days later, become new Xenobots that look and move just like themselves; and then these new Xenobots can go out, find cells, and build copies of themselves.

Kriegman et al. show that clusters of cells, if freed from a developing organism, can similarly find and combine loose cells into clusters that look and move like they do, and that this ability does not have to be specifically evolved or introduced by genetic manipulation. Image credit: Kriegman et al., doi: 10.1073/pnas.2112672118.
“In a Xenopus laevis frog, these embryonic cells would develop into skin. They would be sitting on the outside of a tadpole, keeping out pathogens and redistributing mucus,” said Professor Michael Levin, director of the Allen Discovery Center at Tufts University.
“But we’re putting them into a novel context. We’re giving them a chance to reimagine their multicellularity.”
“People have thought for quite a long time that we’ve worked out all the ways that life can reproduce or replicate,” added Dr. Douglas Blackiston, the senior scientist at Tufts University.
“But this is something that’s never been observed before.”
In their earlier experiments, the authors were amazed that Xenobots could be designed to achieve simple tasks.
Now they are stunned that these biological objects will spontaneously replicate.
“We have the full, unaltered frog genome, but it gave no hint that these cells can work together on this new task, of gathering and then compressing separated cells into working self-copies,” Professor Levin said.
“These are frog cells replicating in a way that is very different from how frogs do it. No animal or plant known to science replicates in this way,” added Dr. Sam Kriegman, a postdoctoral researcher at Tuft’s Allen Center and Harvard University’s Wyss Institute for Biologically Inspired Engineering.
https://youtu.be/aBYtBXaxsOw
On its own, the Xenobot parent, made of some 3,000 cells, forms a sphere.
“These can make children but then the system normally dies out after that. It’s very hard, actually, to get the system to keep reproducing. But with an artificial intelligence program working on the Deep Green supercomputer cluster at Vermont Advanced Computing Core, an evolutionary algorithm was able to test billions of body shapes in simulation — triangles, squares, pyramids, starfish — to find ones that allowed the cells to be more effective at the motion-based ‘kinematic replication’ reported in the new research,” Dr. Kriegman said.
“We asked a supercomputer to figure out how to adjust the shape of the initial parents, and the AI came up with some strange designs after months of chugging away, including one that resembled Pac-Man.”
Kinematic replication is well-known at the level of molecules — but it has never been observed before at the scale of whole cells or organisms.
“We’ve discovered that there is this previously unknown space within organisms, or living systems, and it’s a vast space,” Professor Bongard said.
“How do we then go about exploring that space? We found Xenobots that walk. We found Xenobots that swim. And now, in this study, we’ve found Xenobots that kinematically replicate. What else is out there?”
The researchers see promise in the research for advancements toward regenerative medicine.
“If we knew how to tell collections of cells to do what we wanted them to do, ultimately, that’s regenerative medicine — that’s the solution to traumatic injury, birth defects, cancer, and aging,” Professor Levin said.
“All of these different problems are here because we don’t know how to predict and control what groups of cells are going to build. Xenobots are a new platform for teaching us.”
The team’s work was published in the Proceedings of the National Academy of Sciences.
http://www.sci-news.com/biology/xenobots-10317.html?utm_source=feedburner&utm_medium=email
Thanks to: http://www.sci-news.com






 Sat Mar 23, 2024 11:33 pm by globalturbo
Sat Mar 23, 2024 11:33 pm by globalturbo

